The Objectives: |
The initial objectives of the water treatment (WP3) workpackage was the following:
From the beginning, electrochemical separations were aimed to play a key role, with capacitive deionization (CDI) identified as an interesting technology for the lake water P recovery application. CDI is a novel technological concept with research and feasibility studies being completed on lab scale. CDI for ion selective separation is not yet commercially avalable and thus electrodialysis (ED) as a more mature technology was investigated as performance benchmark. Considering CDI, it was chosen to focus the research efforts on achieving P selectivity from a simple cell configuration with one-compartment cell and without use of ion exchange membranes. The reason was to develop a robust system, where selectivity are achieved from electrode surface caoting and modification. Due to low TRL level of CDI, it was not possible within the timeframe of the rePair to develop a system with capacity to be part of the full scale lake restoration at Lake Ormstrup. For this purpose, sand filtration of the sediment dewatering reject water was used, and research was done to provide the sand filter with P removal ability by mixing in iron oxide sludge from water works. Sand filtration is a very mature technology and this concept could readily be tested in larger scale. To close the loop, P revovery from the generated concentrates were investigated with calcium based chemical precipitations. For more details please refere to the following description. |
The Results: |
The highlights of the workpackage findings are the following:
|
Participants: AAU (JM and MLC) ; DTU (LMO); SDU (KR), AU (AMS)
Description
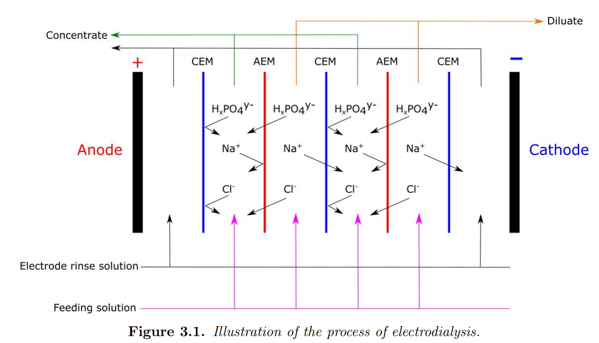
Recovery of P and nitrogen from the flocculated and dewatered lake sediment reject water (WP2) presents a challenge with respect to the expected low concentrations (< 100 µg/L orthophosphate ; < 200 µg/L TP ; < 10 mg/L N-NH4+) that puts high requirements on the efficiencies of the individual and combined treatment processes to achieve an overall cost-beneficial solution. These low nutrient and P concentrations maybe challenging for P removal technologies, these low levels of P can still stimulate excess algae growth and therefore it’s likely the nutrient content is too high in the reject water from the dewatering process to be discharged directly back to the lake. In that case, technologies are needed to generate a concentrated P-rich stream of reduced flow. P can be efficiently precipitated or adsorbed to a solid material to generate a product with fertilizer properties. In WP3, two electric potential-driven technologies, electrodialysis (ED) and capacitive deionization (CDI) will be studied and used for this purpose. ED and CDI has been chosen due to the possibility to achieve sufficient high process efficiencies at low concentrations and since the driving force of the separation is electric potential driven transport through a membrane or electro-adsorption on a surface, and as such may match a treatment concept driven by sustainable electricity production. The clean water effluents of the treated water will be returned to the lake. ED is a mature technology considering other wastewater treatment and brackish water desalination processes (Gurreri et al. 2020) and has also on lab-scale been demonstrated on P removal, but within wastewater applications (Rotta et al. 2019; Kedwell et al. 2019). It has not been demonstrated on lake restoration. The same applies to CDI, even though only few full-scale installations exist (Huang et al. 2014). The main attractive feature of CDI towards lake restoration applications is the enhanced mass transport at low concentration facilitated by the electrostatic attraction that improves the adsorption efficiency compared to pollutant capture and removal by traditional adsorbents (Suss et al. 2015). Selective electrostatic adsorption of P is a major challenge as the process will be in competition with the adsorption of chloride and other naturally present anions. A major factor is the adsorption affinity of the specific electrode material used. A main scientific contribution of this WP is to generate increased fundamental knowledge on how electrode material characteristics and properties influence adsorption mechanisms and affinities.
The experiences obtained in WP3 will in parallel be used to explore a robust treatment concept aimed at low flow continuous treatment of lake bottom water. This application is considered part of potential long-term management of lakes after the sediment dredging and treatment operation and to be used on lakes where sediment removal is not strictly needed.
The concentration technologies produce a P-rich concentrate, from where P is to be recovered. The same applies from the reject water of the electrodialytic sediment treatment (WP4), the hydrothermal treatment of the dewatered sediment in WP5 that generates an aqueous effluent rich in P, and the electroosmotic dewatering (WP6). In WP3, research on precipitation and adsorption technologies will consider the performance of different cheap and sustainable precipitation reagents and adsorbent materials to selectively recover P from these concentrates with the aim of generating a product with fertilizer properties, when adsorption capacity is exhausted. The suggested fertilizer products will be tested in WP7. In focus will be the production of calcium phosphate and struvite products that will provide the benchmark on which more novel minerals, such as vivanite that can be magnetically separated, can be compared. A task in this WP is also to search the literature and scientific networks for state-of-the-art adsorbent materials that may be relevant to test in WP8. Literature review on all technologies explored in the WP is embedded in the tasks presented below to ensure that choices and plans are taken and developed on the current state-of–the-art.
Activity
Task 3.1: Test of electrodialytic reject water polishing and concentrating
Electrodialysis is a commercially available technology that, considering the low ionic strength of the water, may provide sufficient efficiencies for concentrating P in a retentate and allow return of the reject water to the lake. The research focus for this particular application will be studying the influence of electrode and membrane selection, electric potential, and feed composition on solid/reject water flow ratio and compositions and scaling and subsequent cleaning strategy.
Task 3.2: Test of capacitive deionization for reject water polishing and concentrating
Capacitive deionization (CDI) is a technology that can remove ions from a water stream using direct current by electro-adsorption of negatively charged anions to the positive electrode (anode) or positively charged cations to the negative electrode (cathode). The technology is promising with respect to process and energy efficiency, but less commercial even though commercial applications do exist. The research focus will be on the influence of electrode materials, current densities and feed composition and properties on the selective rejection of phosphorous and the combination of the CDI principle with ion-exchange membranes in the so-called membrane CDI (MCDI) process that enhance the selective separation efficiency but increase complexity. When sorption capacity is reached, desorption by discharge is needed that generates a concentrate and another research focus will be to elucidate the reversibility of the adsorption/desorption process.
On the CDI/MCDI technology, collaboration with a profiled international research lab will be engaged and external stays of the PhD student will be planned early in the project to facilitate fast progress within this subject. Identification of the right research environment to approach has been initiated.
Task 3.3: Test of phosphorus recovery by precipitation
Precipitation of P by calcium, ammonium and magnesium to produce solid calcium phosphate and struvite are benchmark technologies that will be tested on the generated concentrates and the mix with reject waters.
A second approach that will be researched is the application of the ViViMAG technology. The EU-funded ViViMAG project develops a magnetic separation technology to recover the iron phosphate mineral vivianite from digested sewage sludge. By the end of 2020, the project should be completed and the technology will be ready for market introduction. We will engage with the vendors and research the use of the technology for this application. Selected mineral products will be tested for fertilizer properties in WP7.
Task 3.4: Test of phosphorus recovery by adsorption
Selective adsorption on filter materials of inorganic and organic nature will be researched. One approach that will be investigated is to use MgxFe layered double hydroxides (LDH). LDHs have demonstrated fast adsorption kinetics and high adsorption efficiency of P in higher concentrations and compose a promising environmentally friendly material for P removal and is a candidate for further investigations. Research focus may be the influence on the feed composition on the adsorption mechanisms of LDHs, the adsorption/desorption selectivity and reversibility, dissolution and reusability of LDHs and possibility of synthesis of LDHs with magnetic properties that ease recovery of the P rich spent sorbent after use.
Task 3.5: Provide state-of-the-art of organic sorbents for p-recovery
We will search the scientific literature and review the most novel research on organic-based sorbents to recover phosphorous. Interesting project ideas generated from this work will be added to the project catalog of WP8 and offered as student projects, thesis projects etc., for further testing on this specific application.
Project structure
JM will be the leader of WP3. The research in the project will be completed by a PhD student (PhD2) hosted at AAU supervised by JM and MLC (AAU) with LMO (DTU) as co-supervisor. The PhD student will focus on the electric potential driven separation processes (electrodialysis and CDI/MCDI) and the use of these technologies for this particular application. Main workplace will be AAU Esbjerg with periods at AAU Aalborg and DTU Lyngby. On the CDI/MCDI technology, collaboration with a profiled international research lab will be engaged and external stay of the PhD student will be planned early in the project to facilitate fast progress within this subject. Identification of the right research environment to approach has been initiated.
Postdoc1 (shared with WP2) will be the lead investigator on the P adsorption and precipitation studies supported by PhD2 with the main workplace at AAU Aalborg. Postdoc supervisor will be MLC, JM from AAU and AMS (AU) as co-supervisor. Collaboration will be engaged with Ulla Gro Nielsen (SDU) on the LDH technology as continuation of already established research activities between AAU and SDU.
Path to commercialization
For lake restoration applications, WP3 will bring ED to TRL6 and CDI to TRL 4. Engagement and collaboration with technology providers are needed to bring the proposed water treatment concept to TRL9 and ready for market. Technology providers do exist, primarily for ED and few for CDI.
Risks associated with work package
A risk associated with WP3 is that the feed water quality from WP2 is insufficient to direct treatment and that further pre-treatment is needed. In this case, filtration will be used to mitigate suspended solids. Another risk is that the P concentration is too low to allow efficient treatment by ED. In this case, more resources will be devoted to the development of the CDI technology.
The Results
Electrodialysis
Electrodialysis achieved complete depletion of phosphate after 4 hours at 15 V, which also was the time where the phosphate begin to concentrate in the concentrate. The 20 hour experiment showed that after the 4 hours, phosphate was continuously concentrated. The lake water experiment showed no sign of fouling. The maximum concentration in the concentrate, as determined in this project, was 1 g/L, without any significant minimization in efficiency and high osmotic effect. Electrodialysis was demonstrated to work as expected, but at a high cost in potential equipment cost and power consumption due to a low current efficiency.
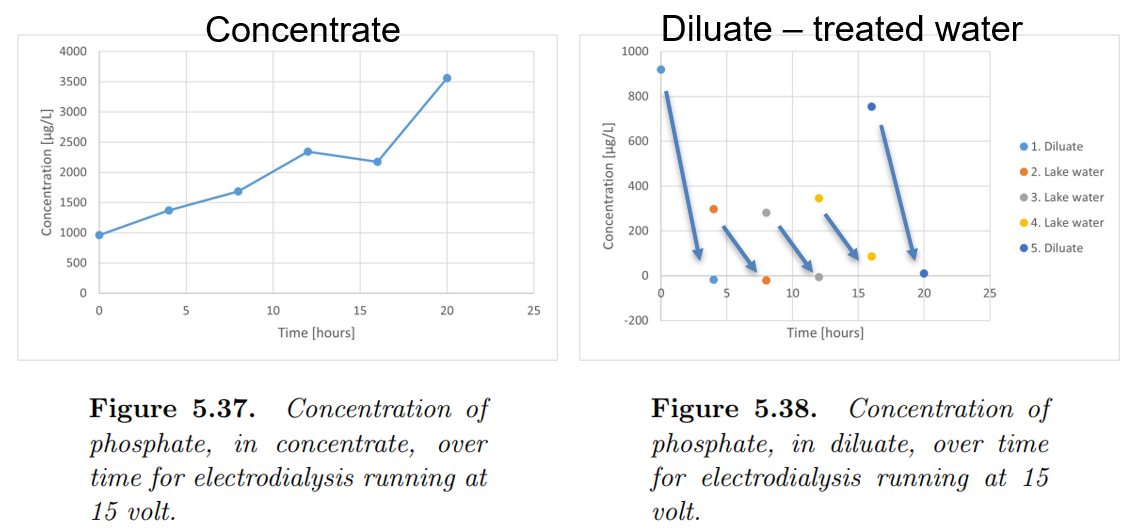
The results on electrodialysis is documented in the master thesis by Stine Louise Bisgaard Rasmussen, Phosphorus Recovery from Lake Water, Master thesis, 2002, Aalborg University.
Capacitive deionisation
The nature of the electrodes used in CDI and ability to selectively and reversible adsorp and desorp phosphates from natural waters with several anions present, is the bottleneck in the further TRL scaling of the technology. It is important that the system is robust, so it was decided to study one compartment 2 electrode systems cells without membranes or reference electrodes that complicates the cell design. We have researched different pathways to achieve selectivity; 1) coating of activated carbon cloth with iron, 2) doping of conductive polymer coatings with phosphate anions, 3) coating of graphite felt with MgAl layered double hydroxides (LDHs), 4) Ca Mg enriched lake sediment biochar electrodes and 5) CDI for P recovery from the anolyte of electrodialytic sediment treatment followed by chemical precipitation.
Iron-loaded activated carbon cloth as CDI electrode material for selective recovery of phosphate
This study investigated the efficacy of oxidised iron-loaded activated carbon cloth (Fe-ACC) for selective recovery of phosphorous. The capacitive deionisation (CDI) technology was employed, for rapid removal of phosphate, with the aim of reducing the reliance on high alkalinity environment for the regeneration of Fe-ACC electrode. Multiple experimental parameters, including applied potential, pH, and co-existing ions, were studied. Additionally, the CDI system was tested on a real water matrix (Lake Ormstrup, Denmark) to elucidate the electrodes’ performance on selective recovery of phosphate. About 69 ±10 % of the adsorbed phosphate were released at pH 12 via pure chemical desorption, which was ~50% higher than that at pH 9. The CDI system successfully demonstrated the selective removal of phosphate from the lake water. It reduced the concentration of phosphate from 1.69 to 0.49 mg/L with a 71% removal efficiency, while the removal percentages of other anions, namely chloride, sulphate, bromide, nitrite, nitrate, and fluoride were 10%, 7%, 1%, 1.5%, 4% and 7%, respectively.
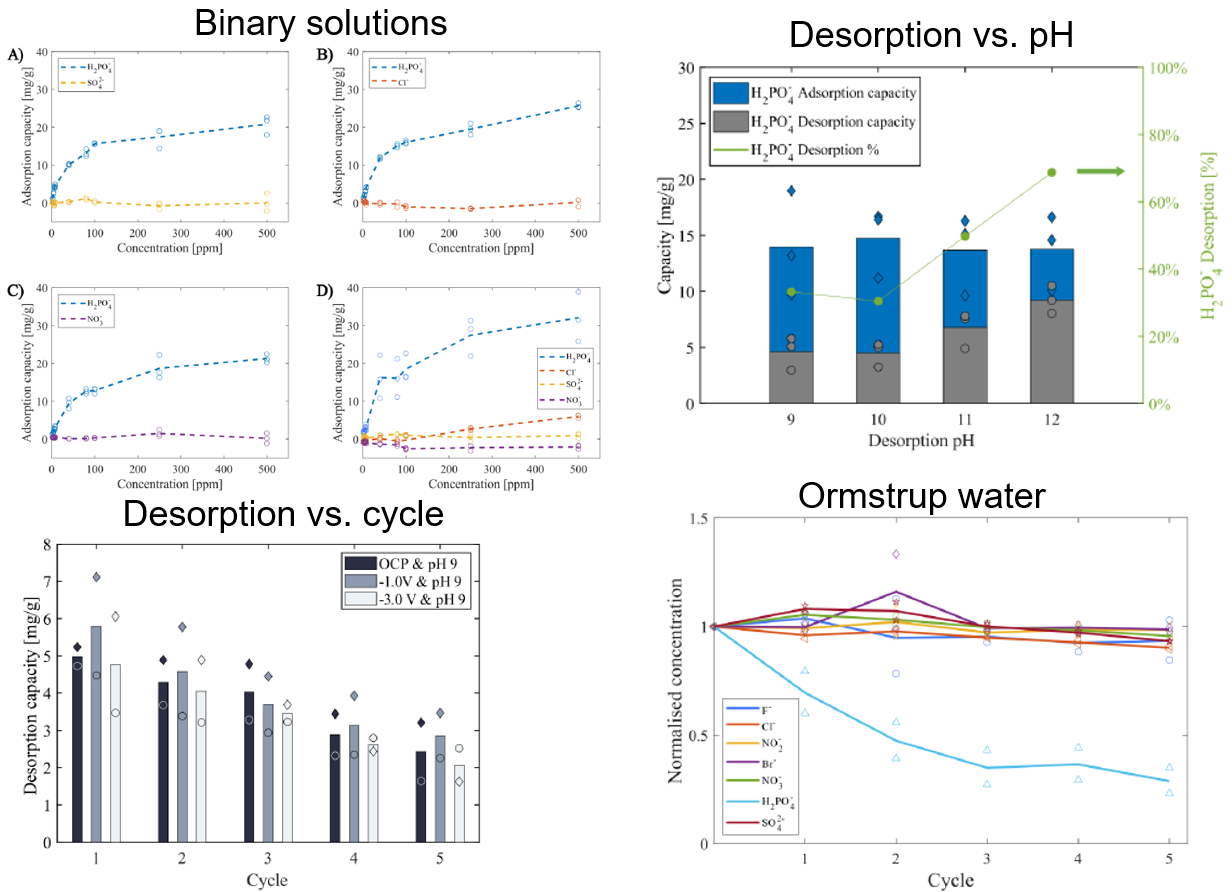
Reference: Sharker, T., Gamaethiralalage, J. G., Qu, Q., Xiao, X., Dykstra, J. E., de Smet, L. C. P. M., & Muff, J. (2024). Iron-loaded activated carbon cloth as CDI electrode material for selective recovery of phosphate. Environmental Science and Pollution Research, 31(55), 63734-63746. https://doi.org/10.1007/s11356-024-35444-7
Effect of dopants in polyaniline-coated capacitive deionization electrodes on anion selectivity
This study explores the viability of employing polyaniline (PAni) coatings doped with different anions in capacitive deionization (CDI) electrodes for ion recovery purposes. Multiple CDI electrodes were fabricated by electrodepositing PAni in the presence of four mineral acids (HCl, HNO3, H2SO4, and H3PO4) to incorporate the corresponding anions into the PAni network. The results highlight the critical role of dopants and polymerization conditions in determining ion adsorption behavior. Notably, the PAni/H2SO4 electrode achieved approximately 20% reduction in chloride adsorption while maintaining consistent sulfate adsorption compared to the control. The behavior of PAni/HCl, PAni/HNO3, and PAni/H3PO4 systems were not as pronounced compared to the PAni/H2SO4 system, under tested parameters, highlighting the dopant-dependent influence of polymerization conditions. Specifically focusing on the PAni/H2SO4 system, various polymerization conditions were investigated, and their ion adsorption characteristics were evaluated using a CDI system. It was observed that reducing the monomer concentration or the polymerization duration can enhance the selective properties of the coated electrode in this specific scenario. Additionally, distinct differences in polymer morphology based on polymerization conditions and dopants were observed, underlining the need for further research to understand the influence of these variables on the morphology of the PAni network and the resulting ion adsorption behavior of the coatings.
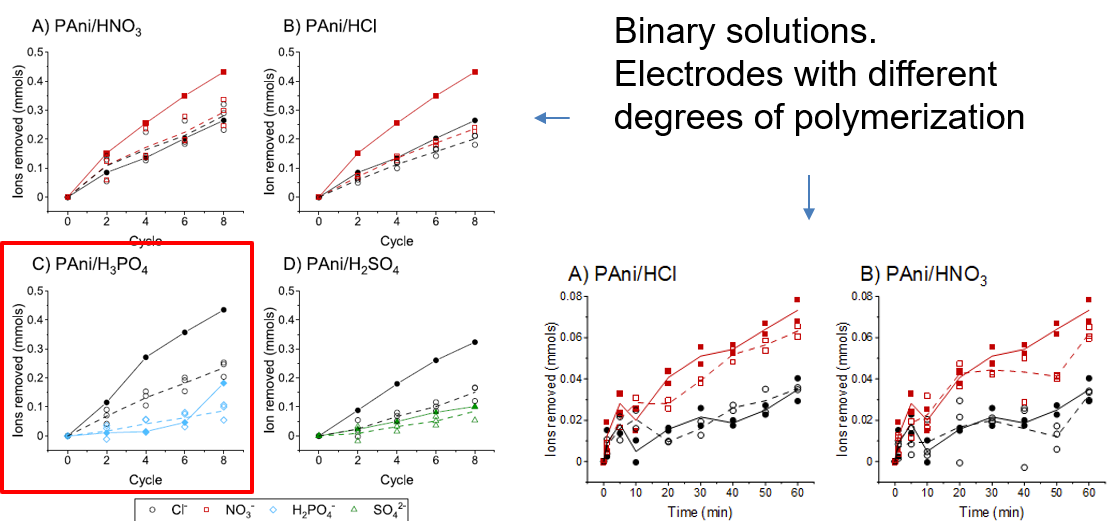
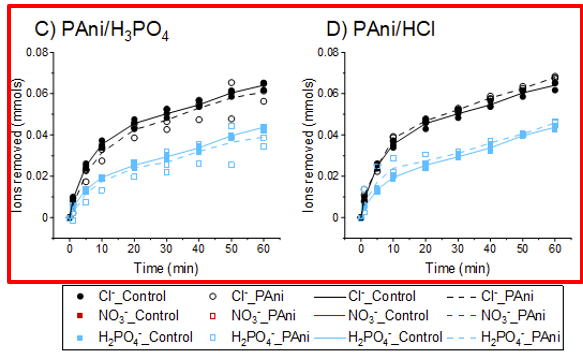
Reference: Gamaethiralalage, J. G., Muff, J., & de Smet, L. C. P. M. (2024). Effect of dopants in polyaniline-coated capacitive deionization electrodes on anion selectivity. Progress in Organic Coatings, 197, Artikel 108854. https://doi.org/10.1016/j.porgcoat.2024.108854
Hybrid capacitive deionization using MgAl-LDHs-coated graphite felt electrodes for phosphate removal
Capacitive deionization (CDI) is a promising technology for selective phosphate removal, though its performance is often constrained by electrode materials. This study developed composite electrodes by integrating MgAl-layered double hydroxides (LDHs) onto conductive oxidized graphite felt (OGF) to improve charge storage and ion selectivity. Two types were tested: CLGF, using commercial nitrate-intercalated LDHs, and LLGF, synthesized with chloride-intercalated LDHs and carbon black for enhanced conductivity. Phosphate adsorption was assessed in both synthetic mixed-ion solutions and real lake sediment dewatering reject water. Kinetic modeling identified chemisorption as the main mechanism, with both LDH-coated electrodes outperforming bare OGF in adsorption and capacitance. LLGF and CLGF showed maximum phosphate removal capacities of ~60 mg/g, while pristine GF and OGF showed negligible ion adsorption capacity. CDI based steady state adsorption capacities stabilized at ~10 mg/g over 5 cycles during phosphate removal from 1.0 mM mixed anions solution. Phosphate-to-sulphate selectivity coefficients were highly time dependent, reaching 2.0 (CLGF) and 4.3 (LLGF) under +1.0 V applied voltage. CLGF removed over 80% of phosphate in reject water at both +1.0 V and open circuit (OC), while LLGF showed moderate phosphate removal (~57%). Energy consumption for the CDI system ranged from 0.03 – 0.25 kWh/m3, within reported CDI benchmarks. Statistical analysis revealed that removal performance was significantly influenced by electrode-time and electrode-voltage interactions rather than individual factors. Overall, this study demonstrates MgAl-LDHs-OGF electrodes as a feasible electrode for lake water P removal with high selectivity towards phosphate over other competing anions.

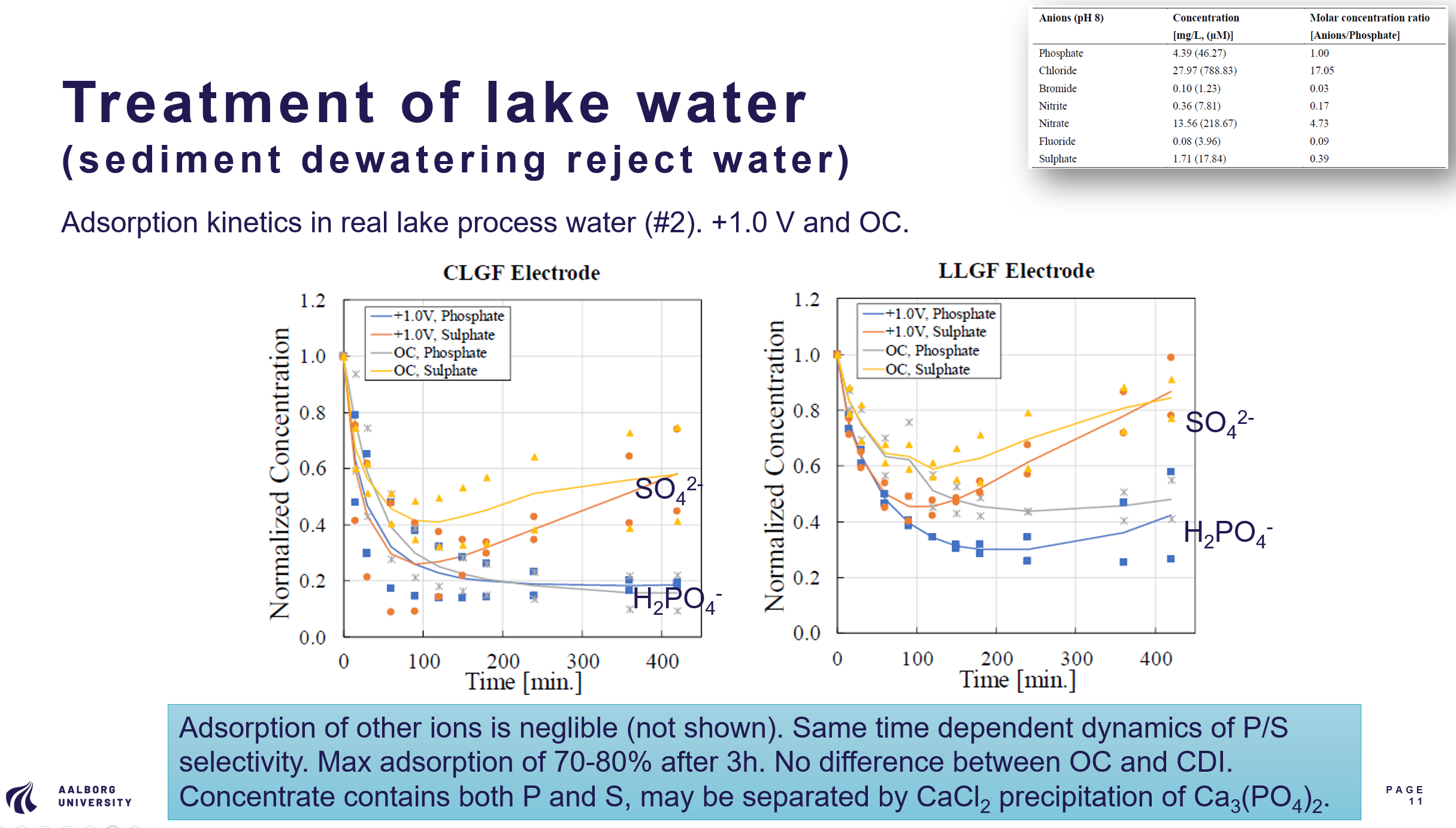
Reference: Sharker, Xiao & Muff (2025), under review for publication
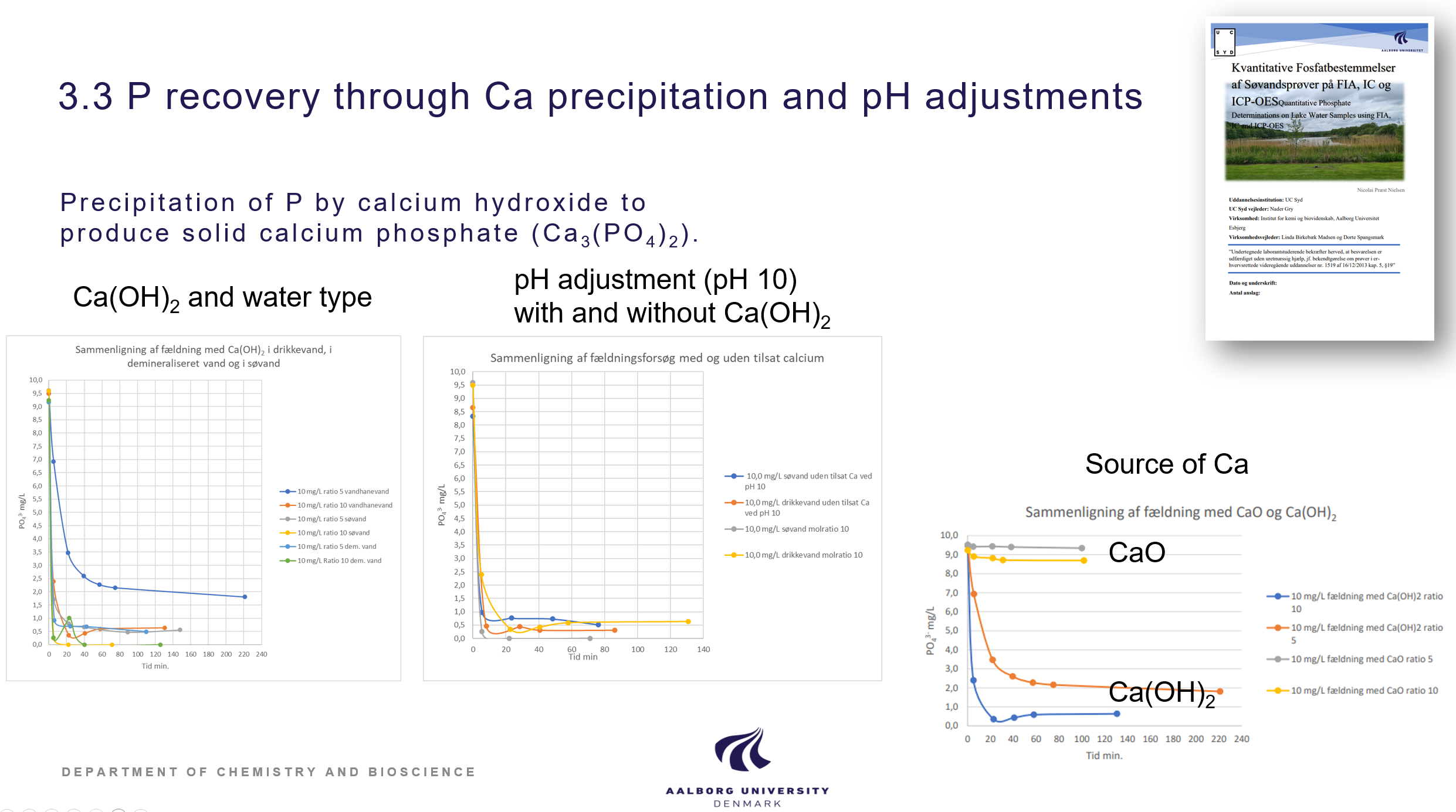
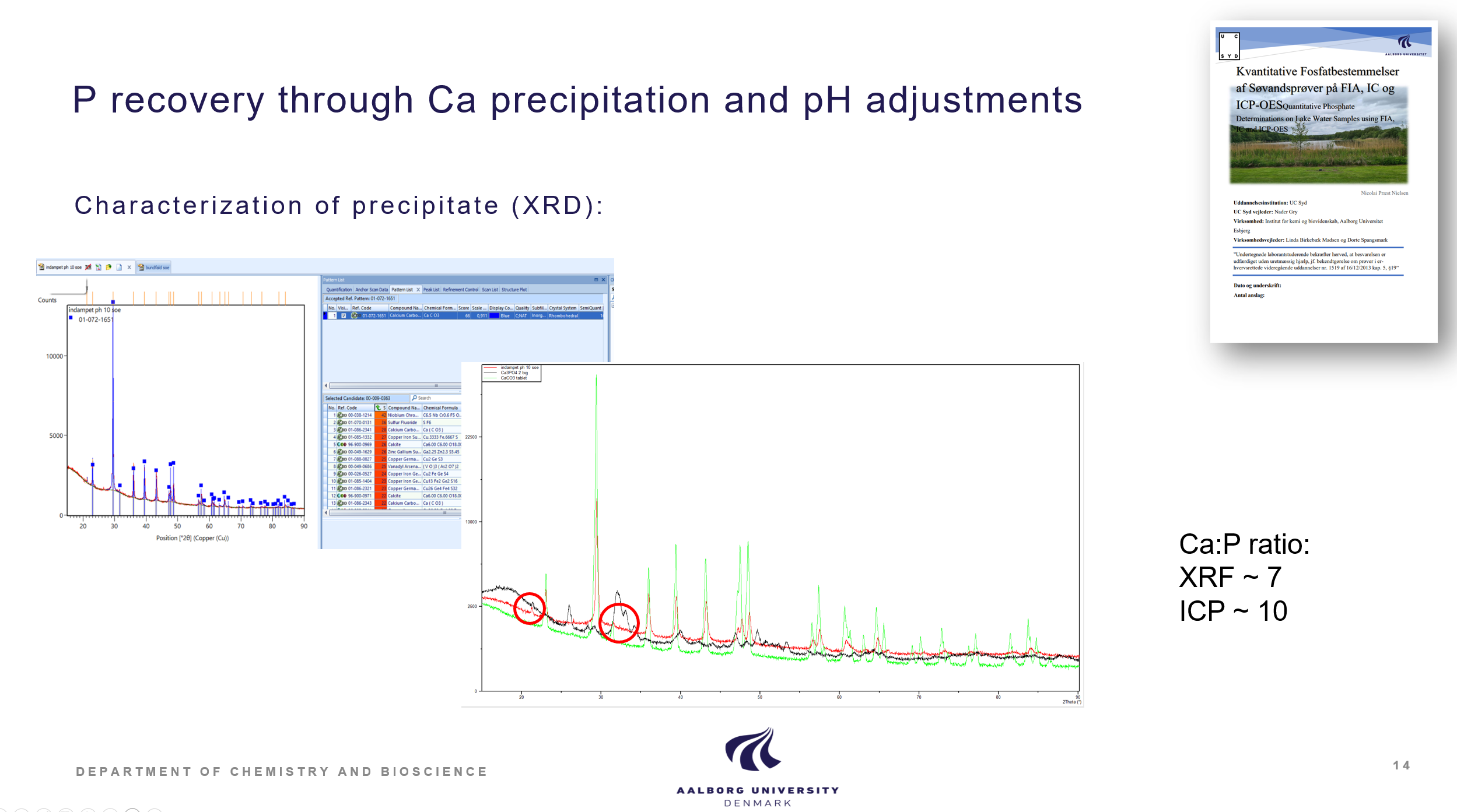
Calcium based precipitation of P
Precipitation of P has been studied by different calcium sources. Calcium hydroxide was demonstrated as a cheap and efficient precipitation reagent for calcium phoshate salts. It was also demonstrated that the lake water contained enough calcium in itself to promote calcium phosphate precipitation just by increasing pH of the concentrates above 10. Calcium based precipitation is documented in the BSc thesis of Edgar Buchaca Tarragona, Phosphorus recovery from contaminated lakes by using calcium salts, 2022, Student report, Aalborg University, and the Laboratory technician final thesis by Nicolai Præst Nielsen, Quantitative Phosphate, Determinations on Lake Water Samples using FIA, IC and ICP-OES, 2022, UC Syd.
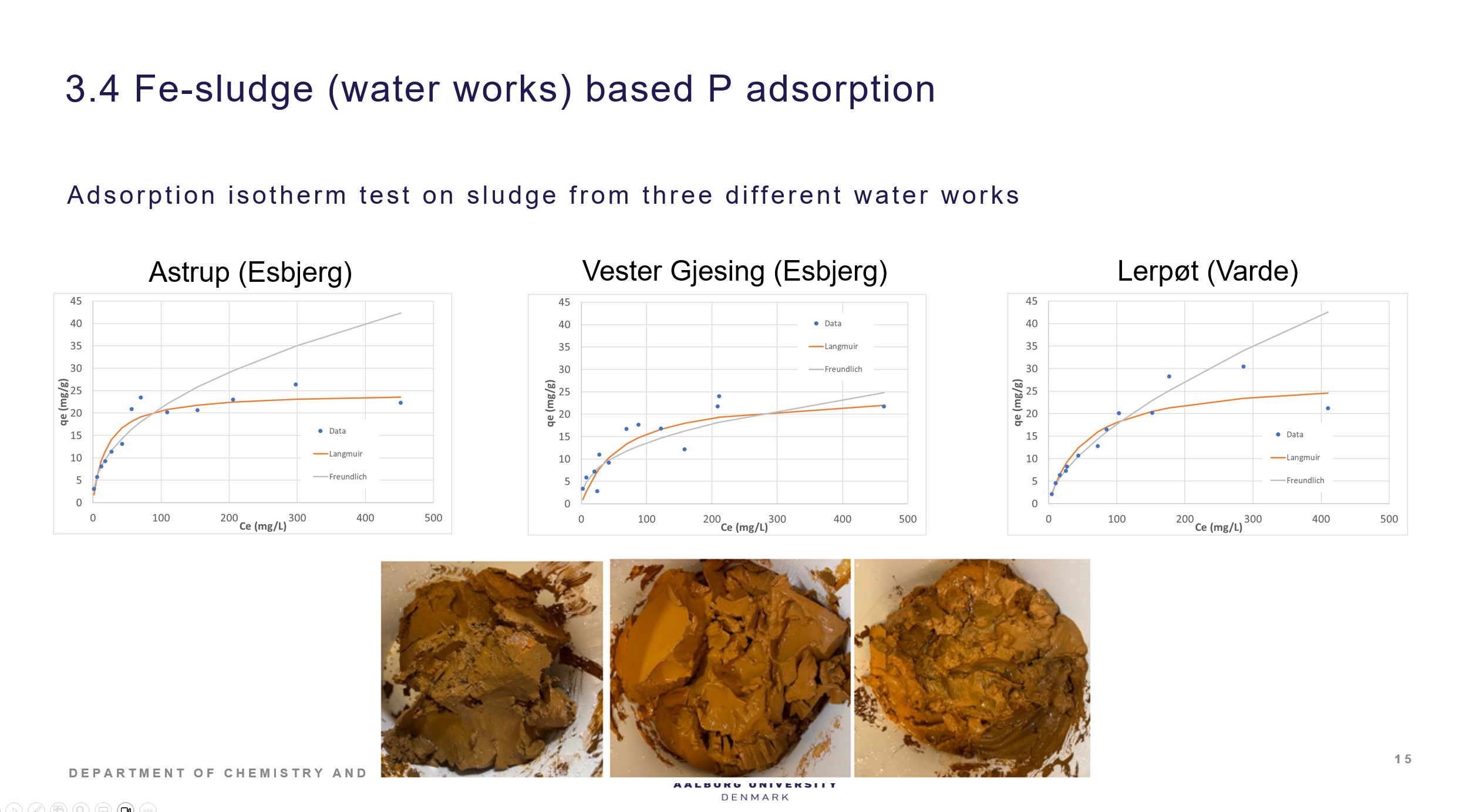
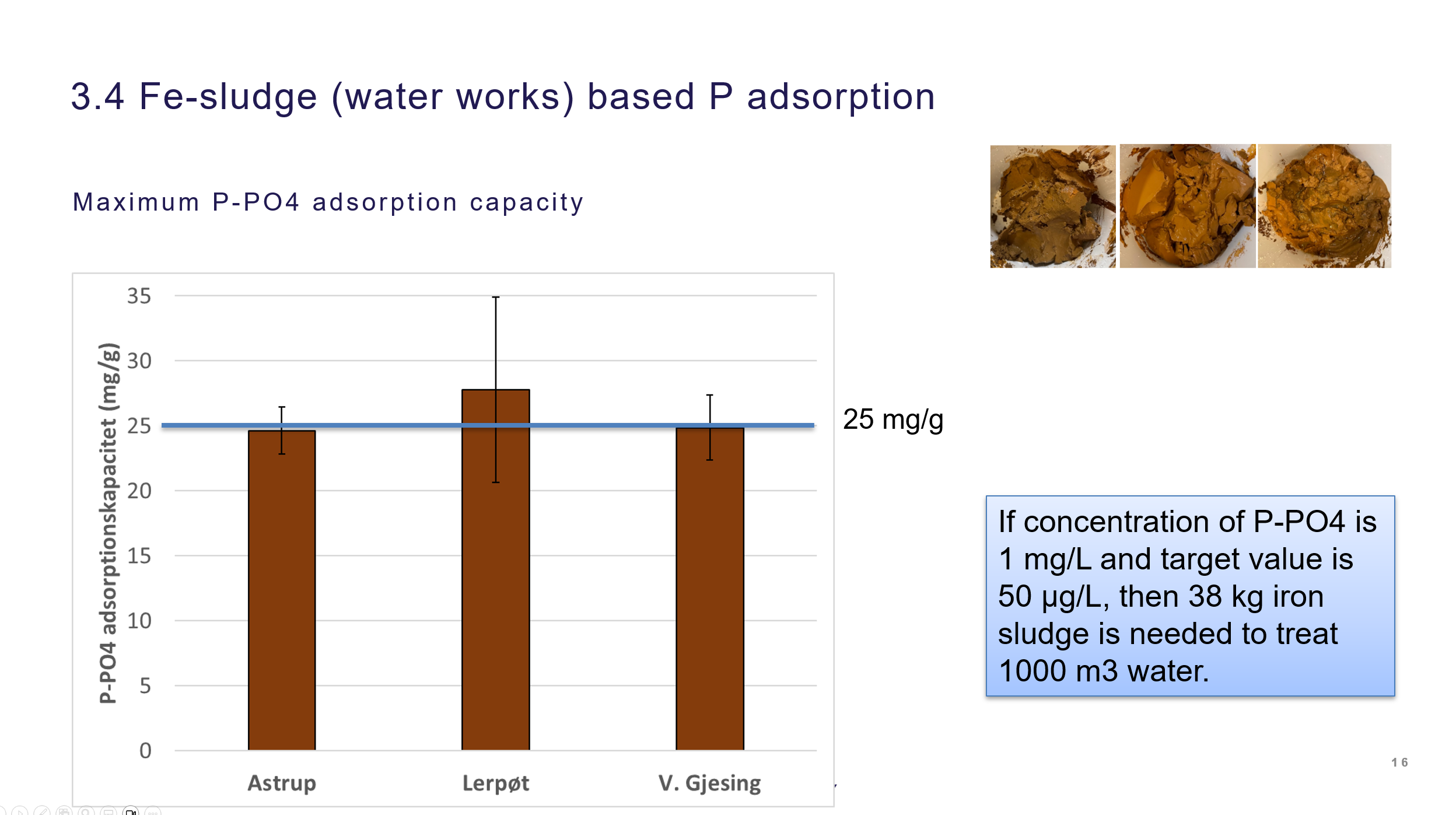
Adsorption of P on iron sludge waste from water works
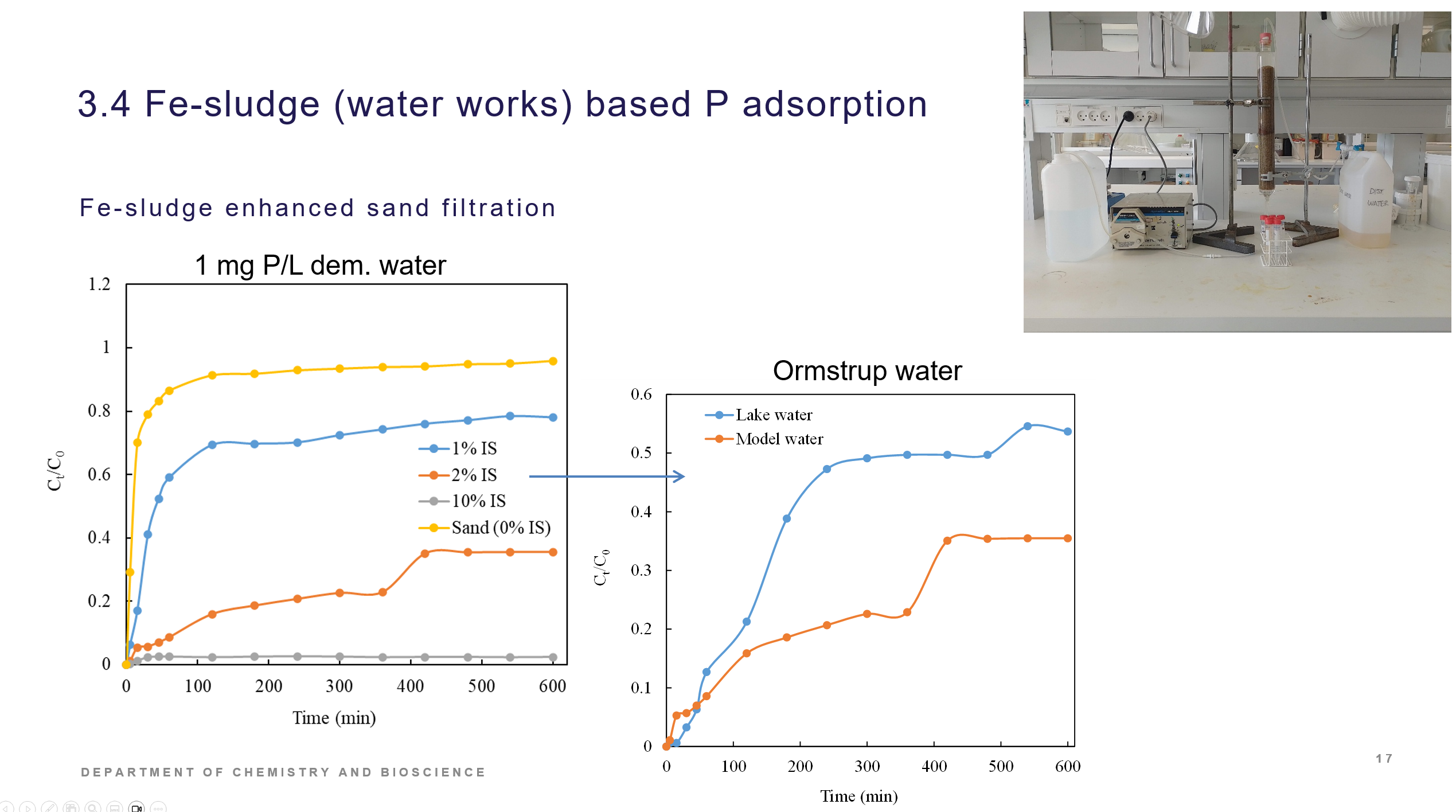
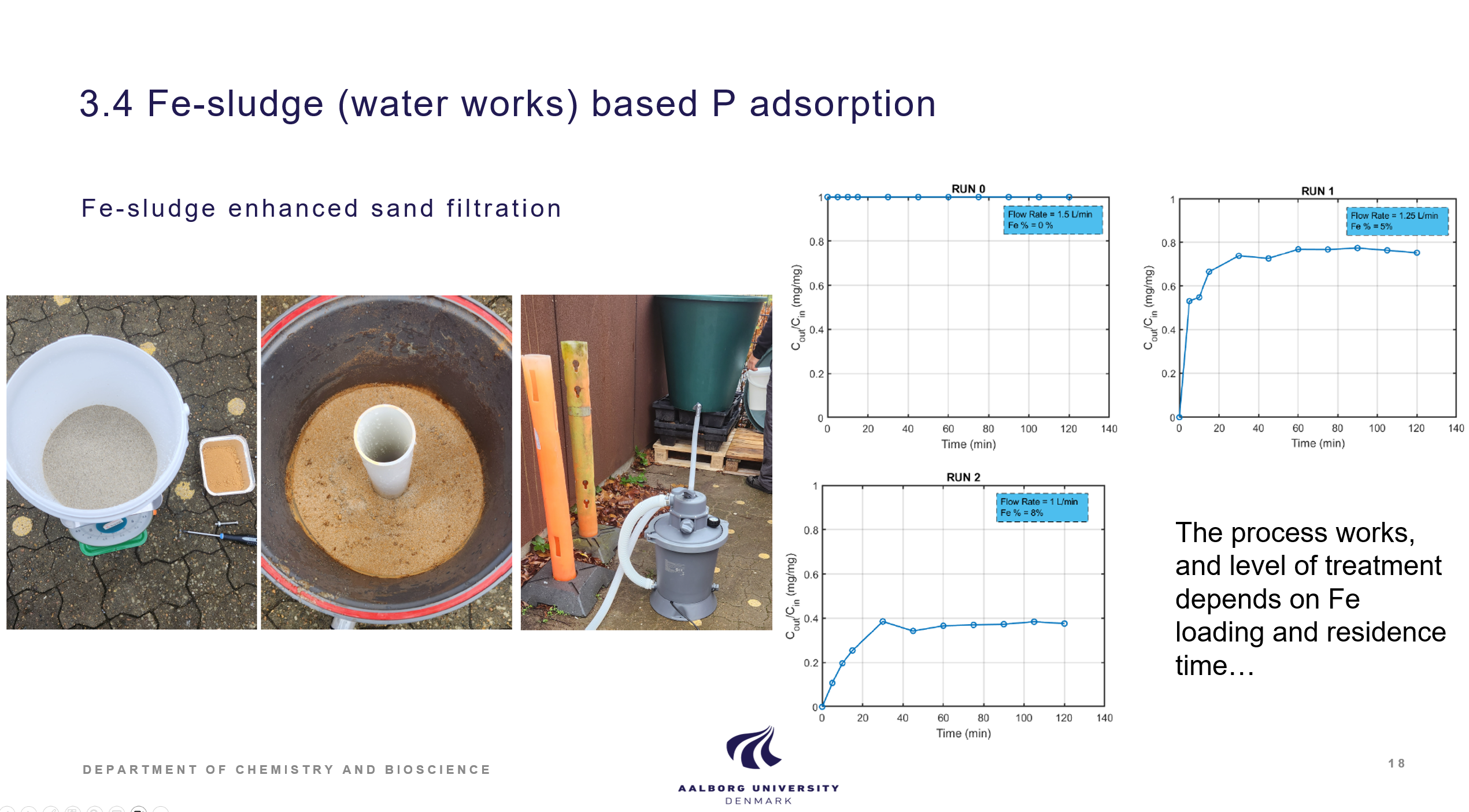
Three different iron sludges was investigated for its P adsorption capacities. They were quite similar and reached max capacities of ca. 25 mg P/ g sludge. Studies on P removal of iron sludge amended sandfilters in lab scale have demonstrated that this approach can lead to sandfilters with P removal ability suitable for scale up. The iron sludge P adsorption is documented in the Laboratory technician thesis by Maria Hansen, Removal of phosphorus in lakes using iron sludge, 2024, UC Syd. The iron sludge amended sandfilter is demonstrated in 5. sem student project (AAU) and in a lab memo report.